Describes the basics of double replacement reactions how to identify them predict the products and balance the chemical equation. A single replacement reaction sometimes called a single displacement reaction is a reaction in which one element is substituted for another element in a compound.
What Is A Double Replacement Reaction In Chemistry Socratic
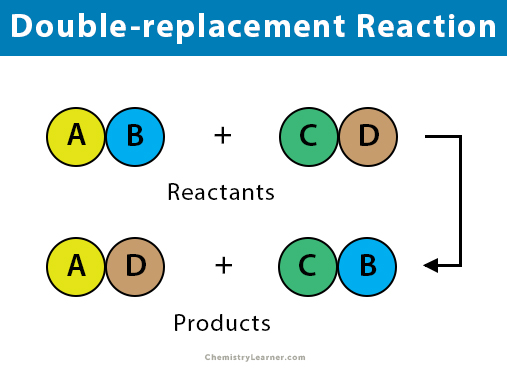
A double replacement reaction is a chemical reaction where two reactant ionic compounds exchange ions to form two new product compounds with the same ionsDouble replacement reactions take the.

. In double replacement reactions the positive ions exchange negative ion partners. A double displacement reaction is also called a double replacement reaction salt metathesis reaction or double decomposition. Two examples are also sho.
There is one other way a double-replacement reaction can happen. Solubility rules are used to predict whether some double-replacement reactions will occur. Many double displacement reactions occur between ionic compounds that are dissolved in water.
Those reactions in which two compounds react by an exchange of ions to form two new compounds are called double displacement reactions. Double replacement reactions are also called double replacement reactions double displacement reactions or metathesis reactions. A precipitation reaction is a double-replacement reaction in which one product is a solid precipitate.
SCICHE515 Double-Replacement Reactions - Chemistry. The starting materials are always pure elements such as a pure zinc metal or hydrogen gas plus an aqueous compound. It is actually a Double replacement reaction when the nitrate and the hydroxidechloride come together to form the compund.
However heat is given off and the pH of both liquids move closer to a. The truefalse questions in this worksheet will help students review the process of a double-replacement reaction. It covers three types.
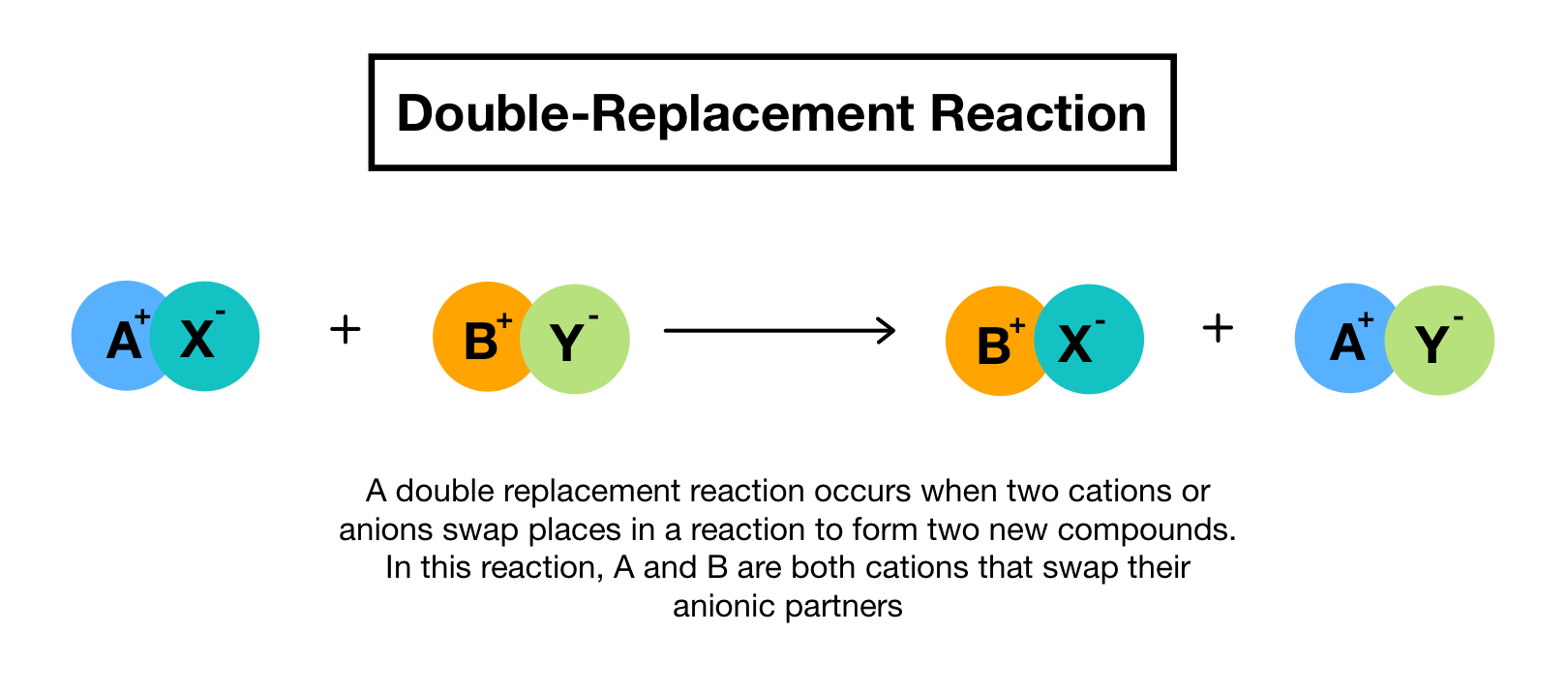
A double replacement reaction is a type of chemical reaction that occurs when two reactants exchange cations or anions to yield two new products. In a double replacement reaction the reactants are always two compounds and the products are always two different compounds. In this printable students also use an equation to answer questions about elements and chemical reactions.
Double replacement reactions often produce precipitates when non-soluble compounds are produced. Thats a sign that the double-replacement reaction is occurring. There are no precipitate or bubbles to show this occurred.
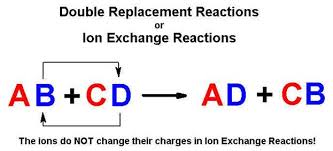
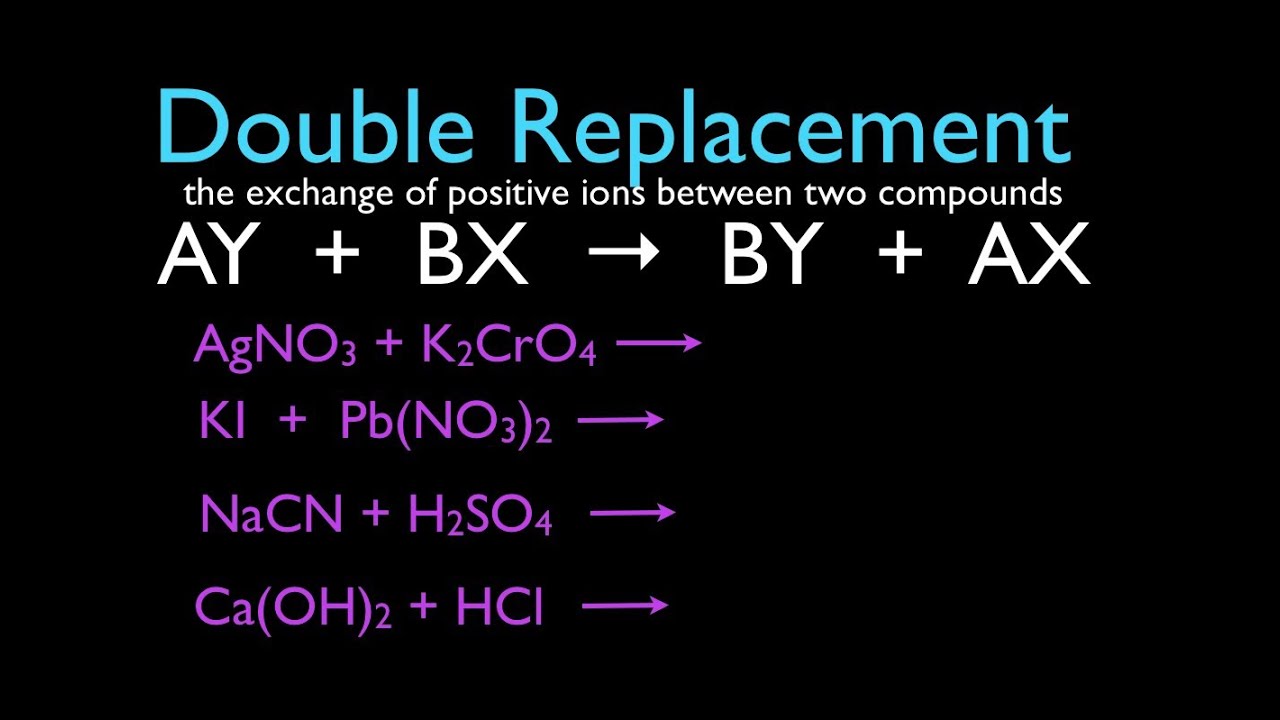
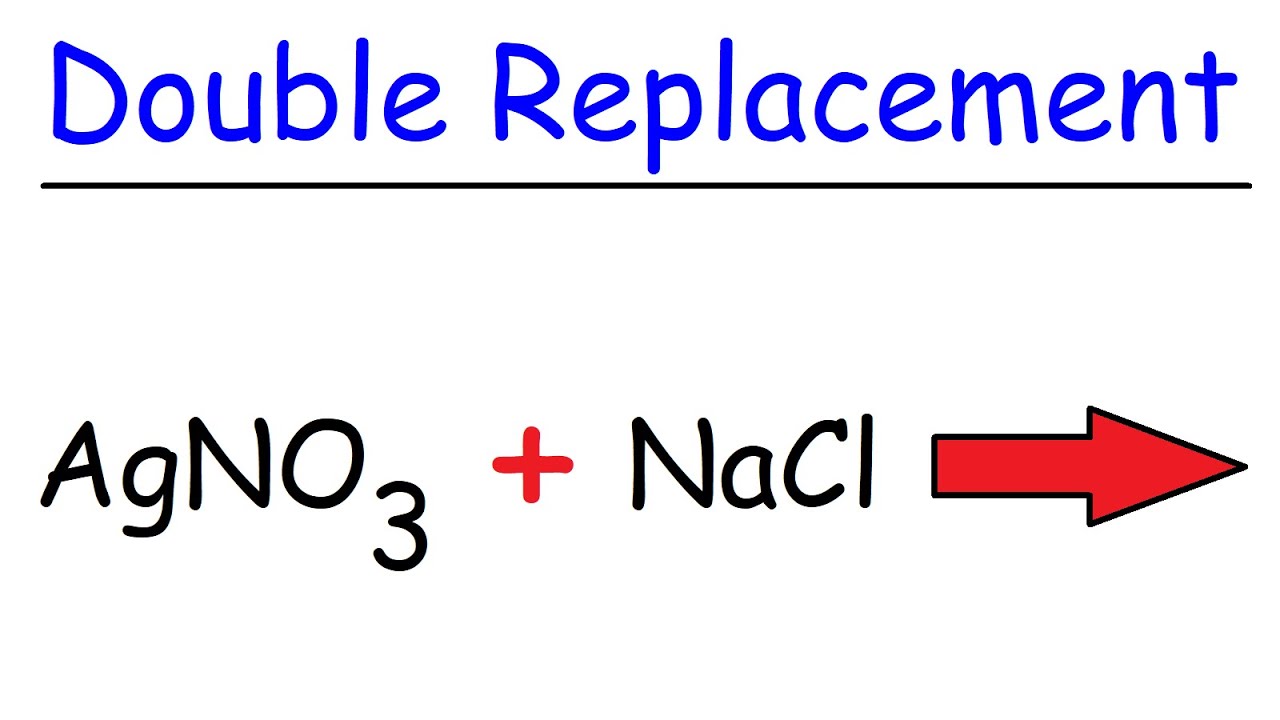
AB CD AD CB Example. AgNO₃aq NaClaq AgCls NaNO₃aq Agaq NO₃aq Naaq Claq AgCls Naaq NO₃aq This is a double replacement reaction because the silver ion and the sodium ion have exchanged partners. The name describes the process very well.
Double replacement reactions take the general form. An aqueous compound formed from single replacement reaction An aqueous compound formed from double replacement reaction A solid formed from a double replacement reaction. When a replacement reaction occurs a new aqueous compound and.
This is simply based on the solubility chart of inorganic compounds. A double replacement reaction is a type of chemical reaction. Select two compounds above and this calculator will predict whether or not the reaction will occur in water.
As a science teacher I can do this kind of thing in my head. H OH- H 2 O. What Is a Double-Replacement Reaction.
The solvent for a double replacement reaction is usually water and the reactants and products are usually ionic compoundsbut they can also be acids or bases. A double replacement reaction will occur if a formation of a precipitate gas or water takes place. A double-replacement reaction exchanges the cations or the anions of two ionic compounds.
A double replacement reaction is. Its when an acid H ions neutralizes hydroxide ions OH- to form water H 2 O. Like single replacement reactions metals always replace metals and nonmetals always replace nonmetals.
This chemistry video tutorial explains how to identify the products of a double replacement reaction from a sentence or word problem. The reaction occurs most often between ionic compounds although technically the bonds formed between the chemical species may be either ionic or covalent in nature. Double replacement sometimes referred to as double displacement reactions are when parts of ionic compounds are switched to form two new ionic compounds.
The name describes the process very well. Gas Ionic molecular compound 3 more precipitate double-displacement reaction double-replacement reaction. A double displacement reaction also known as a double replacement reaction or metathesis is a type of chemical reaction where two compounds react and the positive ions cation and the negative.
Two replacements take place in. In a double replacement reaction two compounds swap atoms forming two different compounds. You can think of the reaction as swapping the cations or the anions but not swapping both since you would end up with the same substances you started with.
The way I think of it since were dealing with ionic compounds is that when I write out a reaction I.
Double Replacement Reactions Definition Examples Expii

Single Replacement Reaction Definition And Examples
Double Replacement Double Displacement Reaction
Chemical Reactions 1 Of 11 Double Replacement Reactions An Explanation Youtube

Double Displacement Reaction Definition Examples Video Lesson Transcript Study Com

Double Replacement Reaction Definition And Examples

Chemical Reactions 2 Of 11 Single Replacement Reactions An Explanation Youtube
0 comments
Post a Comment